After a stroke, many people struggle to find the right words — especially verbs, the engine of every sentence. Imagine wanting to say "I'm cooking" but the word cooking simply won't come. This condition, known as aphasia, affects roughly one-third of stroke survivors, and verb retrieval is often the hardest ability to recover. Our new Cortex study tested whether a gentle form of brain stimulation — transcranial direct current stimulation (tDCS) — can boost verb-naming therapy and, crucially, which brain structures make the difference between a good and a poor response.
What is tDCS?
tDCS delivers a weak electrical current (2 milliamps) through two sponge electrodes placed on the scalp. The positive electrode (anode) slightly increases the excitability of the cortical area underneath, making neurons more likely to fire. Think of it as gently turning up the volume on the brain region you want to engage. When paired with intensive speech therapy, this extra excitability can help the brain relearn connections that the stroke disrupted.

An overview of the tDCS mechanism: from electrode placement over the left inferior frontal gyrus (IFG) to the cascade of neural effects that lead to improved verb retrieval. The right panel highlights the key anatomical insight — preserved frontal regions, particularly the frontal pole, predict better outcomes, while posterior damage is associated with poorer response. Infographic based on: Ilardi, C.R., Federico, G., et al. (2026). Enhancing verb naming with anodal tDCS over the left inferior frontal gyrus. Cortex, 198, 92–109.
Study design
We enrolled 32 individuals with chronic post-stroke aphasia — all at least six months after a left-hemisphere stroke — in a double-blind, randomized, sham-controlled crossover trial. Every participant received both the real stimulation and a "placebo" version (the device switches off after 30 seconds), separated by a one-month washout. Neither the participant, the therapist, nor the experimenter knew which condition was being delivered on any given day.
During each condition, participants completed 10 daily sessions of tDCS over the left IFG combined with a structured verb-naming task: they watched short video clips of everyday actions (e.g., playing guitar, pouring water) and were asked to name the action aloud. Performance was assessed at baseline, immediately after the 10-day block, and one month later.

Schematic of the crossover design. Each participant underwent both conditions — anodal tDCS and sham — in counterbalanced order, separated by a 30-day washout. Verb naming was assessed at three time points per arm. Infographic based on: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
The task
The verb-naming task was carefully designed. Each participant was shown 100 action video clips personalised to their specific deficits — only actions they had failed to name during a screening phase were included. Before each video, a fixation cross appeared for 800 milliseconds. Participants then had 15 seconds to name the action. If they could not, the correct word appeared on screen and they read it aloud, providing an additional learning opportunity.
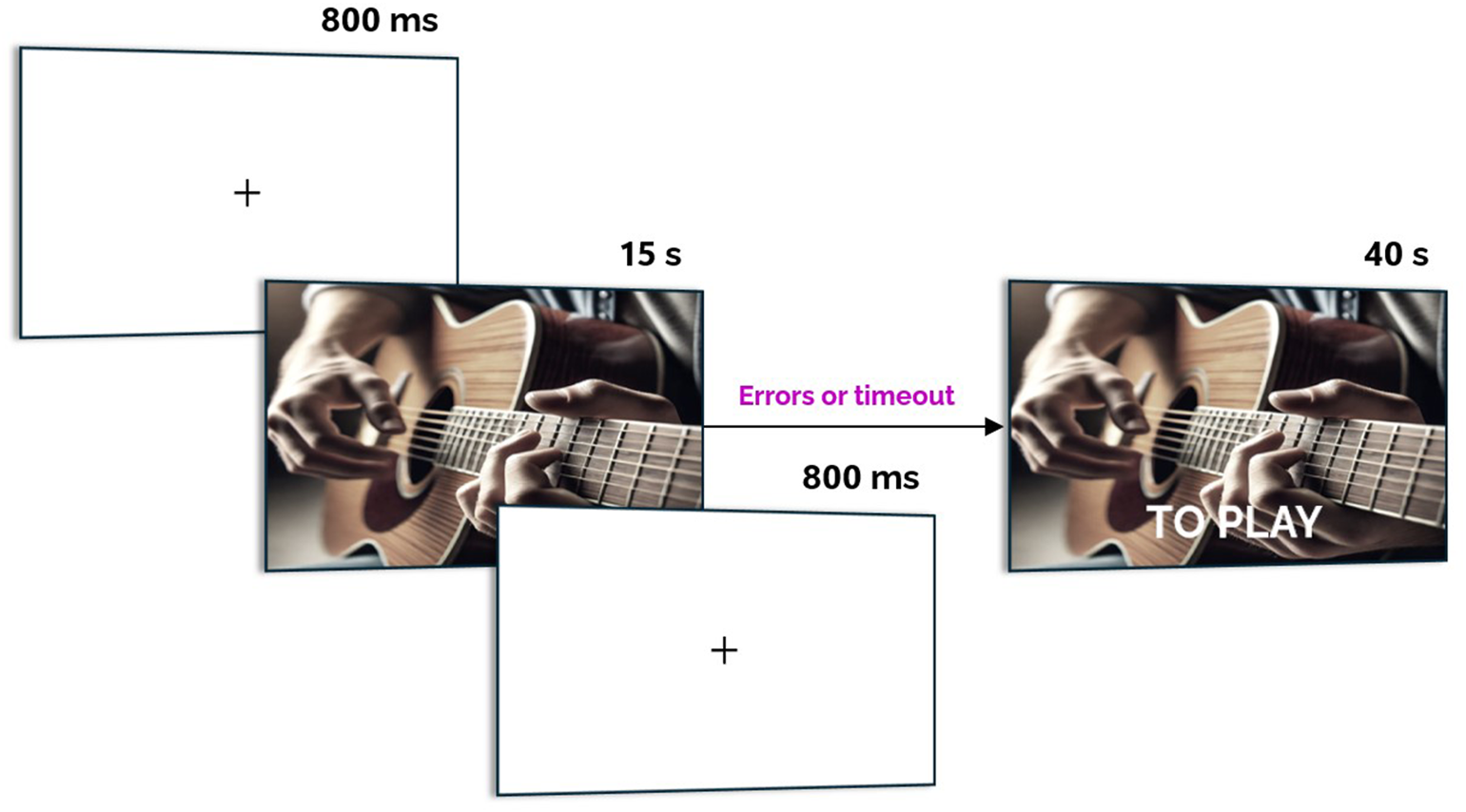
Overview of the action-naming task. Participants watched short video clips showing everyday actions and had to name them aloud. If the response was incorrect or timed out, the correct verb appeared on screen. Adapted from: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
Behavioural results
The results were striking. While both conditions led to some improvement — reflecting the benefit of intensive speech therapy on its own — the anodal tDCS condition produced dramatically larger gains. Every single participant showed greater improvement during real stimulation compared with sham. Performance jumped from an average of around 15% correct at baseline to over 55% after 10 days of anodal tDCS, compared with about 35% in the sham condition. The effect sizes were huge (Cohen's drm = 1.31 at post-treatment; 2.01 at follow-up).
Critically, the gains were sustained one month after the end of treatment. In the sham condition, performance dropped significantly during the same period, whereas in the tDCS condition it remained stable — confirming that the stimulation does more than produce a temporary boost: it promotes lasting neural reorganisation.

Summary of the main behavioural results. All participants improved more under anodal tDCS than sham, with large effect sizes sustained at the 30-day follow-up. The frontal pole emerged as the sole significant anatomical predictor of tDCS response. Infographic based on: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
Transfer to untrained tasks
An important question in rehabilitation is whether improvements on the trained task generalise to other language abilities. We found that tDCS benefits extended to standardised language tests — including picture description, noun naming, and verb naming subtests from the EL-II battery — with moderate effect sizes. This transfer is particularly meaningful for individuals in the chronic phase of stroke recovery, where even modest gains in spontaneous speech can significantly improve daily communication.
Where the lesion matters
Not all brain damage is equal when it comes to predicting who will benefit most from tDCS. We used voxel-based lesion-symptom mapping (VLSM) to link the location of each patient's stroke lesion to their treatment response. Two patterns emerged clearly.
Preserved frontal areas — especially the frontal pole and the middle frontal gyrus — were associated with greater improvement. Conversely, damage to posterior regions — such as the temporal pole, angular gyrus, planum temporale, and superior temporal gyrus — correlated with poorer outcomes. A regression analysis confirmed that the frontal pole was the sole significant predictor of tDCS response, even after controlling for total lesion volume.
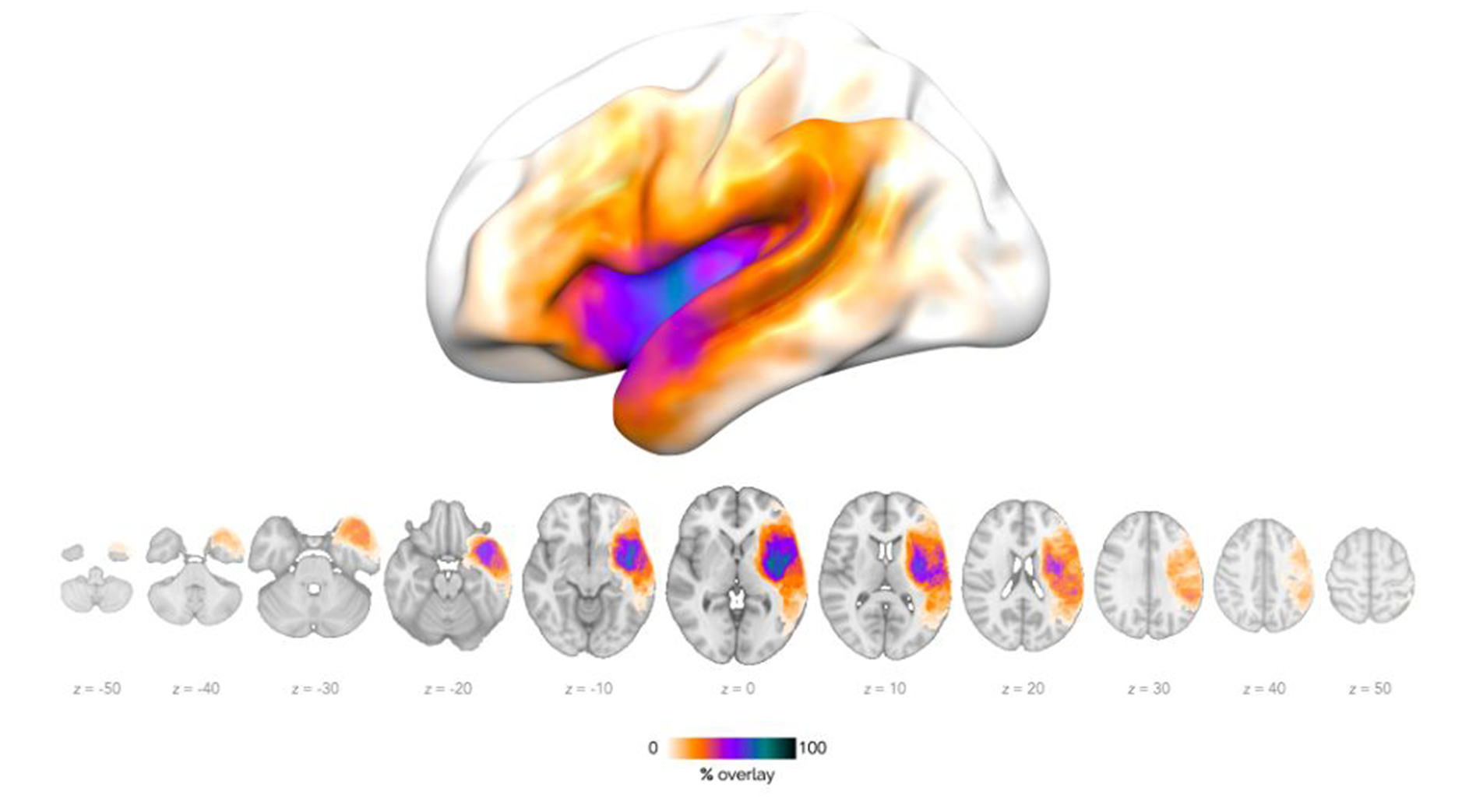
Lesion overlap map across participants, overlaid on the MNI152 standard brain template. Warmer colours (white/orange) indicate voxels affected in a lower number of participants, while cooler colours (purple/black) denote higher lesion overlap. The most frequently affected areas include the left perisylvian region, the insula, the inferior parietal lobule, and the frontal operculum. Adapted from: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
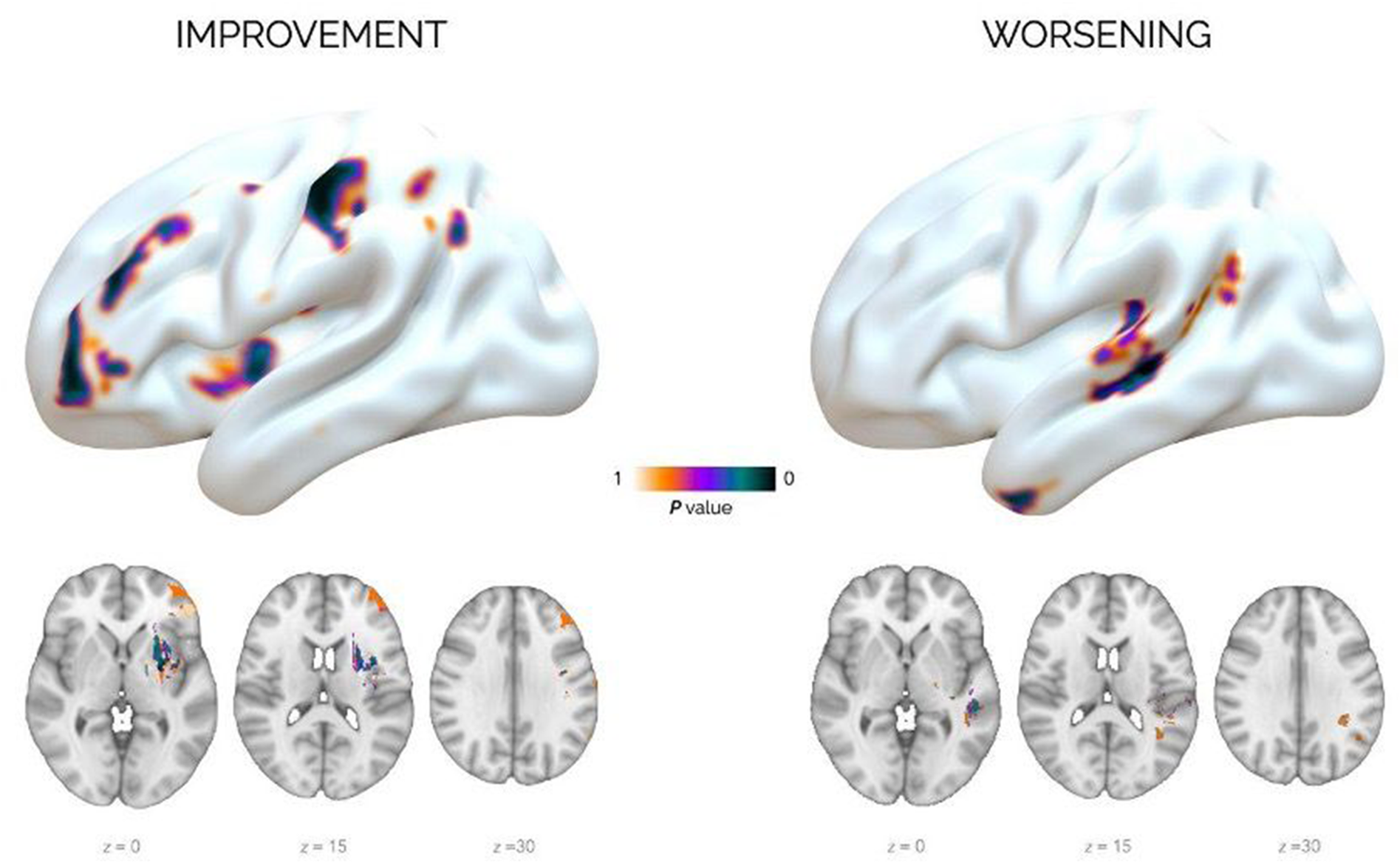
Voxel-wise lesion analysis on verb naming following anodal tDCS. The left panel (Improvement) shows brain areas where tissue preservation was associated with greater benefit; the right panel (Worsening) highlights regions where lesion damage correlated with performance decline. Results are visualised on 3D brain surfaces and axial slices. Adapted from: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
White matter pathways
The brain's regions do not work in isolation — they communicate via bundles of nerve fibres called white matter tracts. To understand which of these pathways were affected in our participants, we projected the group lesion map onto a probabilistic white matter atlas.
The results revealed a clear dissociation. Posterior and dorsal tracts — including the left arcuate fasciculus, superior longitudinal fasciculus (SLF), inferior longitudinal fasciculus (ILF), and inferior fronto-occipital fasciculus (IFOF) — showed high lesion overlap and were associated with reduced treatment benefit. In contrast, anterior tracts — the uncinate fasciculus and the frontal aslant tract — were largely preserved. These anterior pathways connect the IFG to the frontal pole, and their preservation may be the structural basis for the stimulation's effectiveness.

A network-based model of tDCS-driven verb recovery. The stimulation site (left IFG) connects to preserved anterior regions (frontal pole, middle frontal gyrus) via intact white matter tracts (uncinate fasciculus, frontal aslant tract). Posterior regions and their connecting pathways were typically damaged by the stroke, explaining why recovery relies on frontal compensation. Infographic based on: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
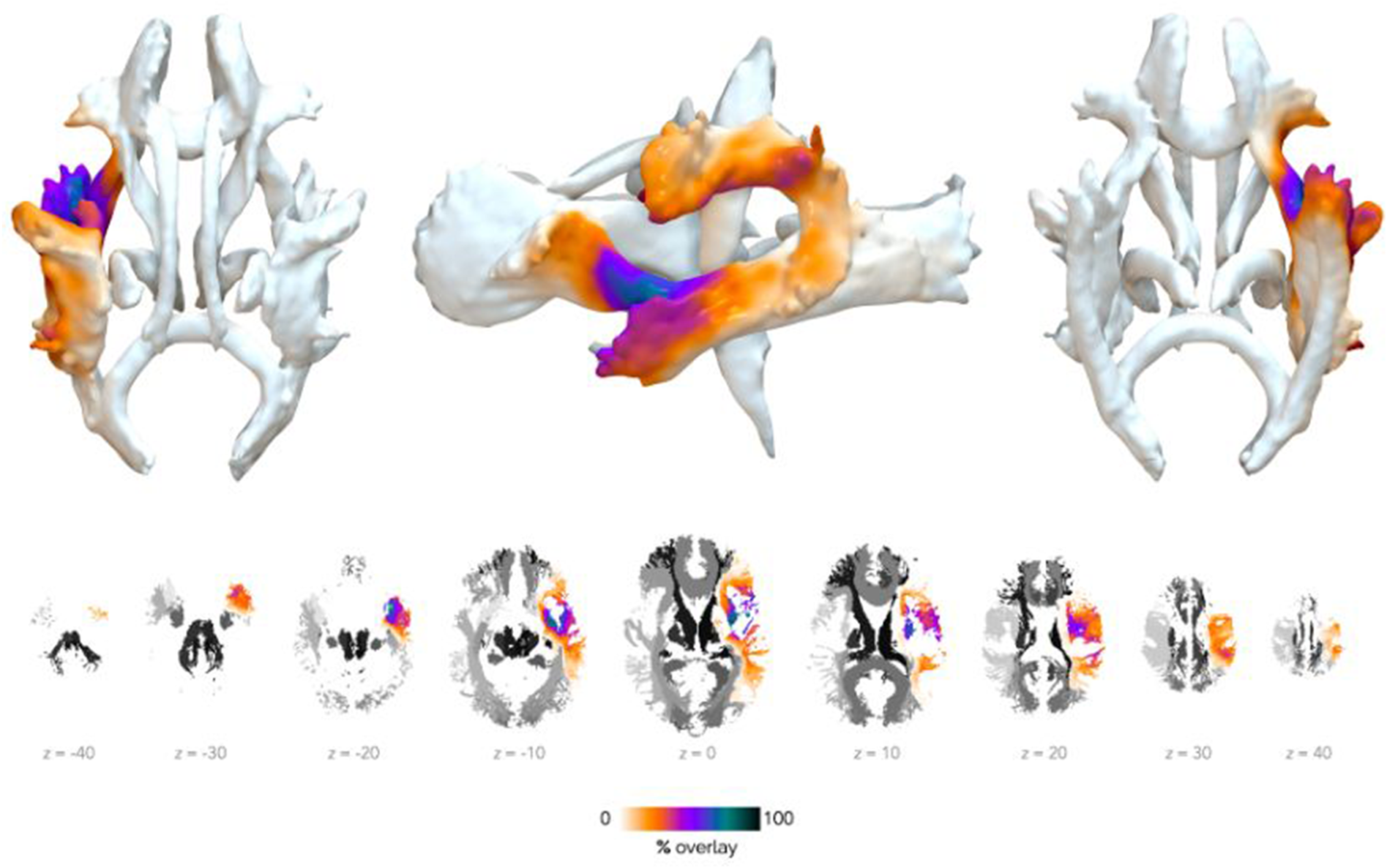
Estimated white matter involvement based on probabilistic atlas mapping. The lesion overlap is projected onto the JHU ICBM-DTI-81 atlas (threshold = 25%). Warmer colours indicate lower lesion probability; cooler colours denote higher overlap. Anterior tracts were relatively spared, while posterior tracts showed extensive damage. Adapted from: Ilardi, C.R., Federico, G., et al. (2026). Cortex, 198, 92–109.
What it means
These findings carry a clear message: tDCS does not work in a vacuum. It amplifies the benefit of structured speech therapy by increasing neural excitability exactly where it is needed — in the left IFG — but its success depends on the integrity of the surrounding network. If the frontal pole and its connections to the IFG are preserved, the brain has the structural scaffolding to support new learning. If those pathways are destroyed, even well-targeted stimulation has less material to work with.
From a clinical perspective, this suggests that lesion anatomy could guide treatment decisions: patients with preserved anterior frontal structures may be ideal candidates for tDCS-augmented therapy, while those with extensive frontal damage may benefit more from alternative approaches. This is a step toward precision neurorehabilitation — tailoring brain stimulation not just to the task, but to the individual patient's anatomy.
Reference
- Ilardi, C.R., Federico, G., Alfano, V., Basso, L., De Luca, M., Caltagirone, C., & Marangolo, P. (2026). Enhancing verb naming with anodal tDCS over the left inferior frontal gyrus: Insights from lesion mapping and structural connectivity estimation. Cortex, 198, 92–109. https://doi.org/10.1016/j.cortex.2026.02.015



